
Nordic Cell Therapy group
Consulting and Expertise in Cell Therapy Development
We do not just advise on cell therapies. We have helped create them.
We know what it takes to bring a therapy to patients, because we have done it.
And we are still doing it.
Nordic Cell Therapy Group Aps, a Danish private limited company
(CVR-nummer 46007441).
Turning cell therapy innovation into reality
We collaborate with innovators, biotechs, big pharma and academic teams to guide cell-based therapies from discovery toward clinical application combining scientific precision, development know-how, and social purpose to move these treatments closer to patients.Nordic Cell Therapy Group — Science guided by experience
Who we are
Nordic Cell Therapy Group unites experienced scientists and industry leaders and and senior consultants dedicated to advancing the field of cell therapy.With decades of combined experience from discovery to commercialization, we bridge the full development continuum, from pluripotent stem cell biology, process and protocol design, and preclinical development to GMP manufacturing and upscaling, regulatory affairs, and market access.Our collective expertise covers every stage of cell therapy development: scientific innovation, process engineering, automation, delivery devices and formulations, non-clinical development, and CMC strategies. We have led and supported programs across multiple therapeutic areas and built productive collaborations between academia, biotech, and industry.We are driven by a shared purpose, to accelerate the translation of cell-based discoveries into safe, effective, and accessible therapies for patients worldwide
our expertise
We provide end-to-end consulting in cell therapy, guiding projects from discovery and process design to clinical and commercial readiness.Our team brings together deep scientific insight and practical development experience across all aspects of cell-based product creation. We support biotech companies, academic groups, and investors in building strong translational strategies, and scalable manufacturing solutions. All geared towards creating value by ensuring reproducibility, robustness, reduced COGS, speed and regulatory compliance.Our expertise spans pluripotent stem cell biology, differentiation and potency assay development, CMC process and analytics, non-clinical validation, regulatory affairs, and commercialization strategy. Whether shaping early research decisions or optimizing late-stage programs, we help our clients turn promising science into tangible therapies for patients.
Our Symbol

The logo of Nordic Cell Therapy Group unites science, medicine, and mythology into one emblem.
At its center lies a cluster of cells evoking the morula, the first form of new life, representing both origin and potential. Surrounding it, the serpent coiled about the staff of Asclepius embodies healing, wisdom, and transformation, while its forward motion conveys progress and protection.In Nordic mythology, the serpent also recalls Jörmungandr, the Midgard Serpent, encircling the earth, a symbol of balance, resilience, and renewal.Together, these elements express our mission: to advance regenerative medicine through knowledge, integrity, and the power of collaboration.The deep green-blue color reflects growth and regeneration, echoing the natural harmony between science and life that defines our work.
Copenhagen, 2300, Denmark
Nordic Cell Therapy Group Aps, a Danish private limited company
(CVR-nummer 46007441).
executive management

Carlos Villaescusa
Co-Founder, CEO & CSOA stem cell biologist and R&D leader with over 20 years of international experience in regenerative medicine, developmental biology, and translational research. At Novo Nordisk Cell Therapy R&D, Carlos served as Head of Department and Research Project Director, guiding multidisciplinary teams and building collaborations that bridged science, CMC, and regulatory strategy. He has led programs across cardiovascular, ophthalmology, hearing loss, and neurodegenerative diseases, advancing pluripotent stem cell–based therapies from discovery to preclinical development. Earlier in his career, he established and directed the Stem Cell and Organoid Core Facility at Karolinska Institutet in Stockholm, supporting both academic and industrial research in organoid and iPSC technologies. His expertise spans pluripotent stem cell biology, cell differentiation and assay development, and translational development of advanced therapy medicinal products (ATMPs).Driven by a commitment to accelerate the translation of cell-based innovations into therapies that improve patients’ lives, combining scientific depth with a pragmatic understanding of development and manufacturing realities.

Michael Wagner Christiansen
Co-Founder, CTOMichael Christiansen is a cell therapy development specialist with over a decade of experience bridging research, process development, and GMP manufacturing in the field of advanced therapies. As Associate Project Director at Novo Nordisk Cell Therapy R&D, Michael has led cross-functional teams responsible for translating laboratory innovation into scalable, compliant, and clinically ready manufacturing processes. His expertise spans 2D & 3D process development, GMP manufacturing and facility requirements, with deep knowledge of regulatory interactions, CMC strategy planning, and technology transfer. Prior to joining Novo Nordisk, he held scientific and engineering roles in both R&D and manufacturing environments.Combines scientific depth with a pragmatic, solutions-driven mindset, helping organizations navigate the complex interface between research and manufacturing to deliver robust, reproducible, and patient-ready cell therapy products.
strategic advisory board

Allan E. Karlsen
Strategic Advisor, Stem Cell Biology & Translational ResearchAllan E. Karlsen has served as Vice President and Head of Stem Cell Research at Novo Nordisk, with over 30 years of leadership experience spanning diabetes biology, regenerative medicine, and translational research. Former Corporate Vice President of the Hagedorn Research Institute and Adjunct Professor at Lund University. Deep expertise in driving innovation from molecular biology to clinical application and building international research collaborations.

Sofia Håkansson Buch
Strategic Advisor, CMC & Manufacturing StrategySofia Håkansson Buch has served as Vice President for Cell Therapy CMC at Novo Nordisk, with nearly 20 years of experience in biopharmaceutical development and manufacturing. Extensive leadership background covering recombinant protein technologies, process development, and production across mammalian and yeast systems. Responsible for establishing and leading global CMC and manufacturing strategies for stem cell–based therapies, bridging research to clinical manufacturing. Deep expertise in process translation, scale-up, and quality systems for advanced therapies.

Tanja Villumsen
Strategic Advisor, Clinical Operations & Supply StrategyTanja Villumsen has served as Head of Clinical Supplies for Cell Therapy CMC at Novo Nordisk, bringing over a decade of expertise at the intersection of clinical operations, regulatory affairs, and pharmaceutical supply strategy. Tanja has spearheaded development of clinical supply chains for stem cell–based therapies, integrating CMC and clinical development to enable first-in-human studies. Her background spans the Danish life science sector and multiple governance roles. She serves on the boards of Pharmadanmark, PFA, and LASA, and contributes to advisory boards at Copenhagen Business School and the University of Copenhagen.
core expert associates

Ida KjærSenior Consultant and CMC specialist with extensive experience in quality systems, raw materials, and late-stage process development for cell-based therapies. Ida has led risk-based raw material management and phase-appropriate GMP strategies, including donor eligibility assessment under Good Tissue Practice (GTP), QMS implementation, and formulation and packaging system design to ensure product integrity and clinical readiness.

Tristan ThwaitesSenior Consultant in cell and gene therapy development with over 15 years of experience advancing genome-edited and stem cell–based products from research to clinical translation. Tristan brings deep expertise in CRISPR, gene editing platform design, and CMC-readiness, having led large cross-functional teams spanning R&D, manufacturing, and regulatory domains in both biotech and pharma. His work bridges scientific innovation and strategic execution, enabling next-generation hypoimmune and allogeneic therapies to reach patients safely and efficiently.

Elizabeth Engenheiro KristiansenSenior Consultant in intellectual property strategy with more than 17 years of experience advising life science organisations on patent protection, portfolio development, and freedom-to-operate for innovative therapeutic platforms. Elizabeth has supported intellectual property strategy across cell and gene therapy, stem-cell engineering, and enabling technologies, guiding patent drafting and prosecution, cross-functional risk assessments, and collaboration agreements between industry and academia to secure long-term competitive positioning.

Chao ShengSenior Consultant with over 17 years of experience across the global ATMP ecosystem, spanning academia, biotech, and CDMO environments. Chao brings strong expertise in ATMP market analysis, product strategy, and value proposition development, combining scientific depth in cell and molecular biology with commercial insight. His work bridges technical innovation and strategic positioning, supporting the successful translation of emerging cell and gene therapy technologies.

Linda B. JensenSenior Consultant with deep expertise in analytical method development and characterization of cell-based therapies. Linda has led the design, qualification, and transfer of analytical assays supporting CMC development, applying Design of Experiments (DoE) and data-driven strategies to ensure robustness and reproducibility. Her work integrates formulation, assay optimization, and quality control, bridging analytical science and product understanding across all development stages.

Joachim HjelmSenior Consultant with broad experience in global drug development and strategy across cell therapies, biologics, peptides, and combination products.
Joachim brings more than two decades of experience defining regulatory pathways and engaging with health authorities in the EU, US, and other key regions worldwide, including senior roles at Novo Nordisk, Ferring, and Lundbeck. His expertise includes CTA/IND requirements, expedited pathways, orphan drug designations, and development-stage guidance across multiple therapeutic areas including endocrinology, cardiology, respiratory, and psychiatry.

Eric ThwaitesSenior Consultant with over 20 years of experience in biopharma and cell-based therapy development, spanning CMC strategy, manufacturing, and global commercialization. Eric played a key role in bringing the world’s first allogeneic mesenchymal stem cell therapy (Alofisel®) to market and has led global manufacturing and supply chain strategy at Takeda and Novo Nordisk. His expertise includes process development, technology transfer, due diligence, and life-cycle management for advanced therapy products.

Kan KanekoConsultant with extensive experience in device and delivery strategies for cell-based therapies. Kan has a strong background in formulation development, combination-product testing, and optimization of administration parameters from preclinical to translational stages. His work bridges the interface between cells, materials, and devices, ensuring robust delivery, stability, and usability of advanced therapy products.

Anna DrozdConsultant with expertise in raw materials strategy and qualification for cell-based therapies.
Anna brings extensive experience in early evaluation, risk assessment, and supplier collaboration for cellular starting materials, excipients, and plastics. Her work bridges material science and regulatory expectations, ensuring phase-appropriate compliance and robust partnerships that strengthen advanced therapy manufacturing.

Peter OverbyConsultant with broad expertise in diabetes, metabolism, and stem cell–based therapies. Peter has extensive hands-on experience with rodent models, preclinical efficacy studies, and translational assessments of novel therapeutic approaches, including cell and device-based transplantation models. His background bridges metabolic disease biology, immunology, and in vivo pharmacology, supporting non-clinical development and cross-functional project execution from discovery to IND-enabling stages.
extended expert network
Beyond the core team, the Nordic Cell Therapy Group is supported by an extended network of over 30 experienced professionals covering the full spectrum of cell therapy development, from discovery and process engineering to clinical, regulatory, and commercial translation. This network includes former industry, academic, and CDMO specialists who can be engaged on demand, allowing Nordic Cell Therapy Group to rapidly assemble tailored teams for each project and provide end-to-end expertise across all stages of advanced therapy development.

Join Our Expert Network !!!
We are always looking to connect with talented professionals who share our passion for advancing cell therapy.
If you have expertise in CMC, regulatory affairs, analytics, clinical translation, or related areas and want to help shape the future of regenerative medicine... we would love to hear from you!!!
Use the contact form to tell us about your background and what motivates you to be part of the consultancy arm of the Nordic Cell Therapy Group.

Equality, Inclusion & Ethical Conduct
At Nordic Cell Therapy Group, we are committed to a respectful and inclusive working environment for all.
We do not tolerate discrimination based on gender, sexual orientation, gender identity, religion, ethnicity, nationality, age, or disability.We value professionalism, integrity, and mutual respect in every collaboration, and we ensure equal opportunities and fair treatment for everyone we work with, internally and externally.As we engage with partners around the world, we uphold universal principles of human dignity, ethical conduct, and responsible science.
Our services
Every project is different. With our consultancy services, we adapt to your needs, helping you turn scientific vision into clinical reality.
Translational Strategy & Program DesignFrom concept to clinical planWe help transform scientific discoveries into structured development programs.
Our team supports early-stage projects in defining translational objectives, critical experiments, and milestones that align with clinical and regulatory expectations.
We bring hands-on experience from multiple cell-therapy programs to ensure each step builds toward a viable therapeutic product.protocol design · pluripotent stem-cell biology · differentiation workflows · potency assays · in vivo pharmacology · biomarker strategy · regulatory scientific advice meetings
Process Development & CMC StrategyBridging science and manufacturingWe guide clients in developing scalable, reproducible, robust, and GMP compliant manufacturing processes, with focus on reducing COGS. Based on the Quality by Design approach, we can help you define your Quality Target Product Profile, Critical Quality Attributes, release specifications, raw material risk assessments, and the overall product risk assessment. We can help design your analytical qualification programs, including writing protocols and reports, and if
needed execute the study on your behalf. Whether your ambitions are fast to first human dose or fast to market, we can guide your strategies accordingly. We ensure that your production platform is ready for clinical and regulatory scrutiny.process design · 2D & 3D upscaling · formulation & cryopreservation · raw-material strategy · QA / QMS · CDMO selection & oversight · delivery device
Non-Clinical & Regulatory GuidanceDe-risking the path to first-in-humanWe design and interpret non-clinical studies that demonstrate safety, potency, and mechanism of action. Our consultants help translate data into regulatory-ready packages and align study plans with global requirements. We also support clients in preparing regulatory documentation and engaging effectively with authorities.study design · histology · pharmacology · CRO selection · IND / CTA strategy · regulatory documentation · biomarker integration
Clinical & Operational SupportFrom the lab to the clinical siteWe help coordinate the first steps of clinical implementation, ensuring alignment between CMC, regulatory, and clinical teams.
Our services include all steps from clinical trial design to clinical supply mapping and coordination, as well as final qualification of the on-site dosing process. We will ensure that your secondary endpoints, stopping rules and follow-up programs are compliant with regulators expectations. We work closely with CDMOs and clinical partners to maintain quality and consistency through every stage.site initiation & dosing support · person-in-plant supervision · due diligence · trial design · clinical supply · risk management · quality monitoring
Intellectual Property & Business DevelopmentTurning science into sustainable impactWe assist companies and academic spin-offs in building partnerships, communicating with investors, and planning for long-term growth.
Our team combines scientific insight with strategic perspective to help position your technology for clinical and commercial success.patents · business strategy · partnerships · market positioning · investor communication
Emerging TechnologiesPreparing for what comes nextInnovation drives progress in cell therapy. We help clients evaluate and integrate new technologies, from genome engineering and device development to automation and data-driven manufacturing, always with quality and compliance in mind.genome editing · automation · device integration · combination products
ready to collaborate?
Let’s accelerate the future of cell therapy together.→ Contact us to discuss how our expertise can support your program
Nordic CelL Therapy R&D Pipeline
Advancing the Next Generation of Cell TherapiesIn parallel with our consulting activities, Nordic Cell Therapy R&D is actively developing proprietary regenerative medicine programs.
Our internal R&D efforts build on years of experience in pluripotent stem cell biology, differentiation, and translational development, with a focus on diseases where cell replacement can bring meaningful patient benefit.While details of our ongoing projects remain confidential for now, our team is pursuing first-in-class cell-based approaches that address areas of high unmet medical need. Each program integrates scientific innovation with pragmatic development planning from process design to non-clinical evaluation and regulatory strategy.Our R&D group currently includes a dedicated team of scientists and project leaders with expertise spanning discovery biology, CMC, and translational development. Together, we aim to transform advanced stem-cell concepts into future therapeutic solutions.Science in motion — advancing tomorrow’s therapies, today
Limbal Stem Cell Deficiency (LSCD) Program
LSCD is a rare but devastating condition in which the limbal epithelial stem cells, responsible for maintaining and regenerating the corneal surface, are lost or dysfunctional. Patients experience chronic pain, recurrent epithelial breakdown, inflammation, and progressive loss of vision. Severe cases, including those affecting both eyes, often leave individuals with no effective long-term treatment options.Advances in regenerative medicine are opening new possibilities for restoring the integrity and function of the ocular surface. At the Nordic Cell Therapy Group, we are committed to translating these possibilities into meaningful therapeutic solutions.
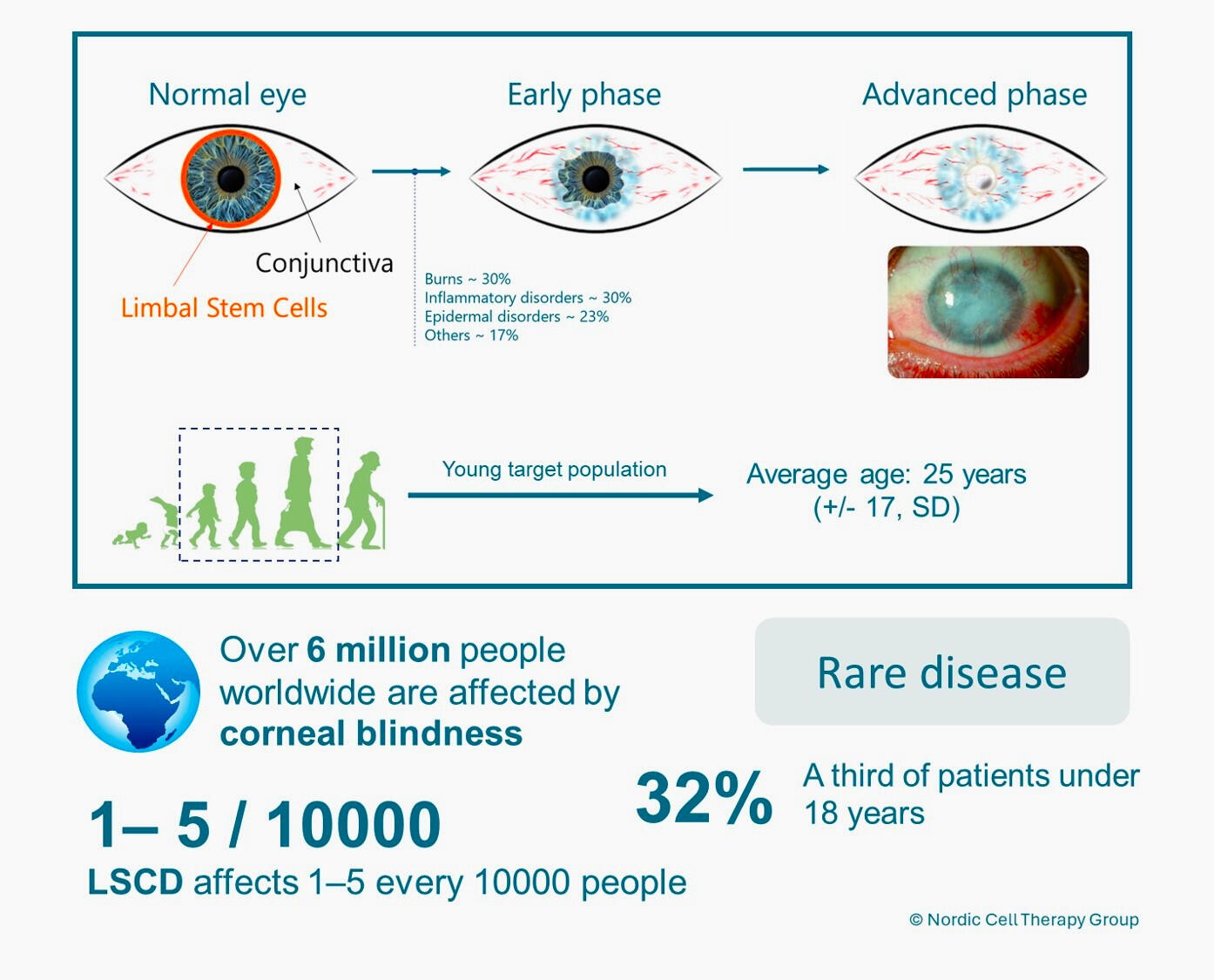
Our ApproachNordic Cell Therapy R&D is developing a next-generation limbal stem cell–based therapy designed to restore corneal epithelial regeneration in patients with LSCD. Our approach builds on three pillars:1. State-of-the-art stem cell biology.
2. Translational expertise across preclinical research, CMC, and regulatory strategy.
3. A rigorous development framework focusing on safety, reproducibility, and scalability.Our LSCD program is currently in pre-clinical development, where we are establishing the foundational scientific work needed to advance toward future clinical translation.
Program StatusThe LSCD program has reached a stage of pre-clinical maturity, supported by ongoing in vitro studies and early exploratory in vivo activities. Future work will focus on:¤ Optimizing the cell therapy product and process.
¤ Advancing translational models.
¤ Preparing for CMC and regulatory readiness.
¤ Building the partnerships required for clinical progression.We pursue this development with a strong commitment to scientific integrity, patient needs, and responsible innovation.
Partner With UsNordic Cell Therapy R&D welcomes collaboration with clinicians, research groups, foundations, and investors who share our vision of enabling new therapeutic options for LSCD.
To explore partnership, scientific collaboration, or support for this program, please contact us.
Further Reading & Resources
R&D Contributors
Our research and development initiatives are supported by a group of experienced scientists who have previously worked together on pluripotent stem cell–based programs.
They bring complementary expertise in cell biology, process development, and translational research, strengthening Nordic Cell Therapy Groups’s ability to design and evaluate next-generation regenerative therapies.

Andreas WronaSenior Scientist with broad experience in cell therapy research and development, combining expertise in stem cell differentiation, proteomics, and image-based analysis. Over eight years in Cell Therapy developing stem cell–derived therapies for ophthalmology and cardiology, with a focus on assay design, biomarker discovery, and translational support.

Xiaogang GuoSenior Scientist specialized in genetic analysis and CMC analytics for cell-based therapies. Xiaogang has led the development of advanced genetic stability and off-target profiling platforms, implementing long-read sequencing, optical genome mapping, and variant interpretation pipelines to ensure the integrity of gene-edited cell products.

Signe FrazierScientist with expertise in neural stem cells, neuronal differentiation, and extracellular vesicles for neurodegenerative disorders. Signe recently completed her PhD with Novo Nordisk and the University of Copenhagen, focusing on neural stem cell–derived exosomes and their therapeutic potential in neurodegeneration.
They collaborate with Nordic Cell Therapy Group on a project basis as independent professionals, contributing voluntarily to our early R&D programs.
Support Our Research
Be Part of the Future of Cell TherapyAt Nordic Cell Therapy Group, we are advancing innovative research programs that aim to make cell-based therapies a reality for patients.
While our consultancy drives progress across the field, our internal R&D arm focuses on developing next-generation regenerative treatments in areas of high unmet medical need.Private contributions and research sponsorships play a vital role in accelerating this work.
Every donation directly supports experimental research, from early laboratory studies to preclinical validation, helping us move promising therapies closer to the clinic.If you share our vision and wish to support one of our research programs, please reach out to discuss how your contribution can make a measurable impact.Together, we can help shape the future of cell therapy. One breakthrough at a time.→ Contact us about research support
Contact
Ready to collaborate?
The path to cell therapy is long. We help you cross it with clarity, experience, and purpose.
Together, we connect science, strategy, and execution to bring therapies closer to patients.

Use the form below to reach out — we’ll get back to you over the next hours.
Copenhagen, 2300, Denmark
Nordic Cell Therapy Group Aps, a Danish private limited company
(CVR-nummer 46007441).
FAQ — Nordic Cell Therapy Group
What types of projects do you support?We focus exclusively on cell therapy. Our consultancy assists biotech companies, start-ups, and academic groups in developing cell-based products, from stem-cell biology and assay development to CMC strategy, non-clinical design, and regulatory preparation. Each collaboration is tailored to the project’s maturity and needs.What is your main goal?Our mission is simple: to accelerate the development of cell therapies for the benefit of patients.
Every project we take on is guided by that purpose, translating scientific innovation into treatments that can make a real impact.Are your consulting and R&D activities separate?Yes. Our consultancy and internal R&D operate independently to avoid conflicts of interest.
The consulting team focuses solely on supporting clients, while our R&D group pursues proprietary regenerative-medicine programs under strict confidentiality and ethical boundaries.How do you work with clients?We start with an introductory conversation to understand your scientific and strategic goals. Based on that, we outline a focused scope of work, timeline, and deliverables. Communication is direct and transparent, from initial discussion to final hand-off.How is your pricing structured?We offer flexible collaboration models: hourly consulting, defined project packages, or retainer arrangements for long-term programs. All terms are agreed in advance, with clear expectations and no hidden costs.Do you sign NDAs?Yes. Protecting your data and intellectual property is fundamental to how we work. We routinely sign Mutual Non-Disclosure Agreements and treat all preliminary discussions as confidential.Do you work only with established companies?Not at all. We frequently collaborate with early-stage ventures and academic groups preparing for translation. Our role is to help transform promising research into development strategies that meet industrial and regulatory standards.Do you raise funding or invest in client projects?Our focus is on scientific and translational development, not financial brokerage.
However, because we actively develop cell therapies ourselves, we are always open to exploring collaborative projects or alternative paths that help promising therapies reach patients.Can I donate or support your research?Yes. In addition to our consultancy, Nordic Cell Therapy Group conducts internal R&D programs in regenerative medicine.
Private contributions and research sponsorships are always welcome to help advance these programs by supporting specific studies that aim to move promising cell therapies closer to patients.As a Danish ApS company, we are not a non-profit organization, and contributions are treated as research sponsorships rather than tax-deductible donations.
All funds received are used exclusively for scientific activities within the designated research programs.If you would like to support our work or learn more about available programs, please contact us directly to discuss how your contribution can make an impact.